Last Line of Defense
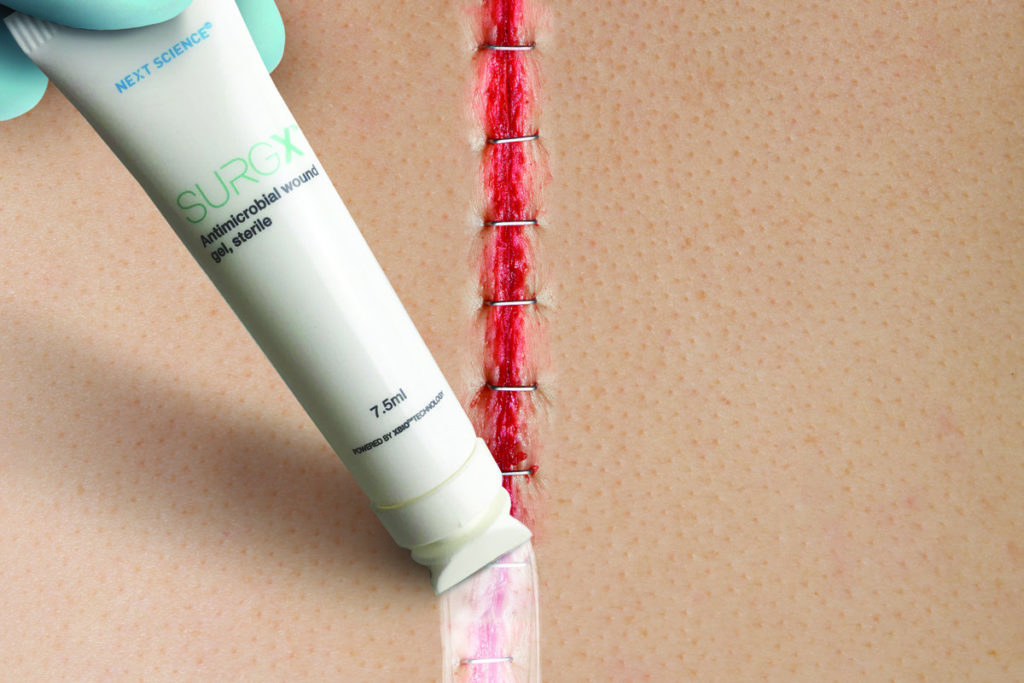
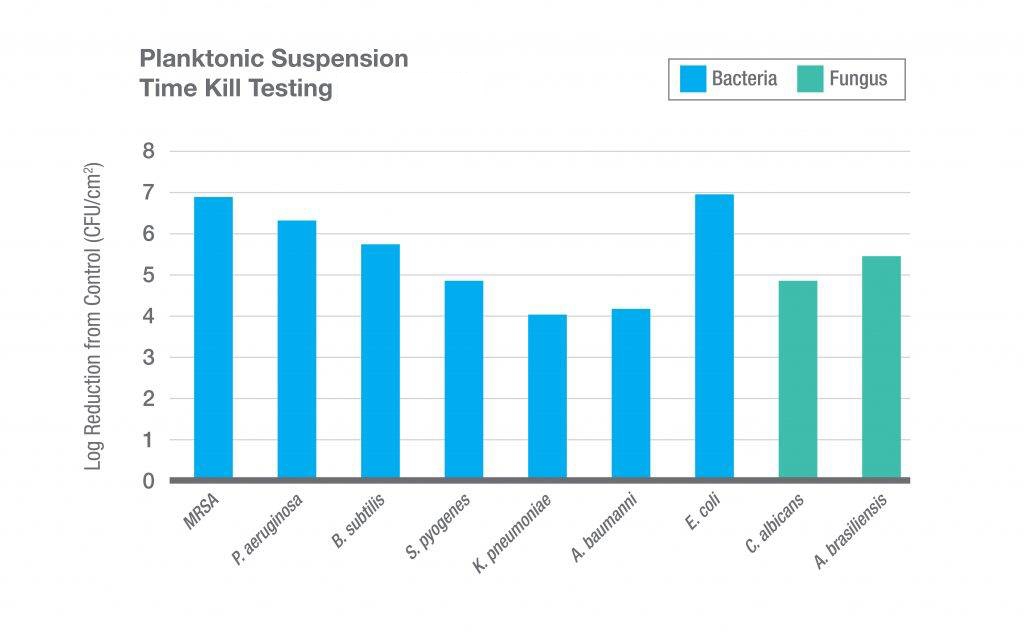
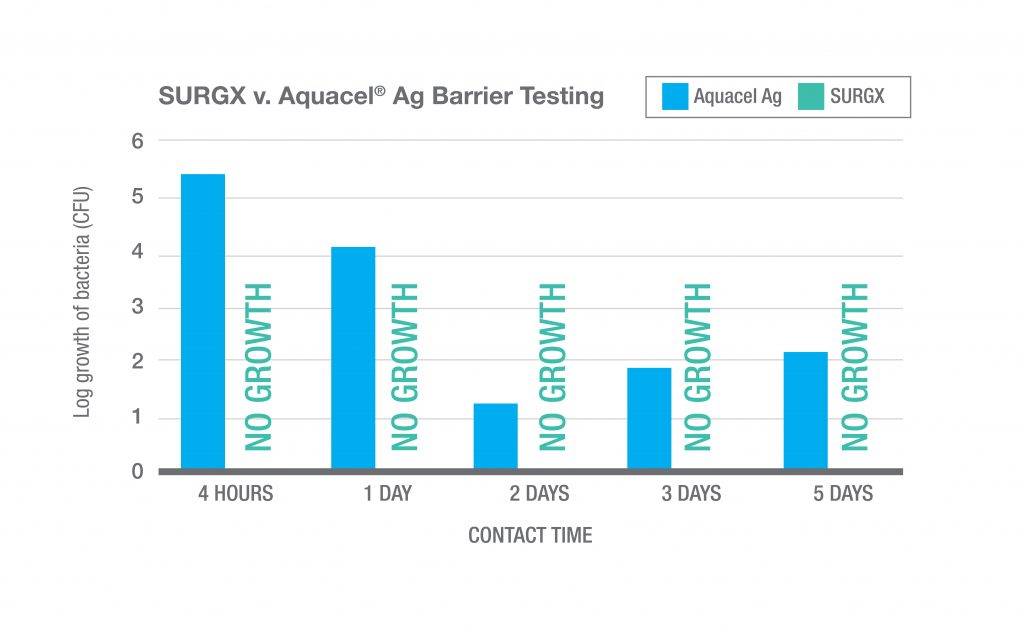
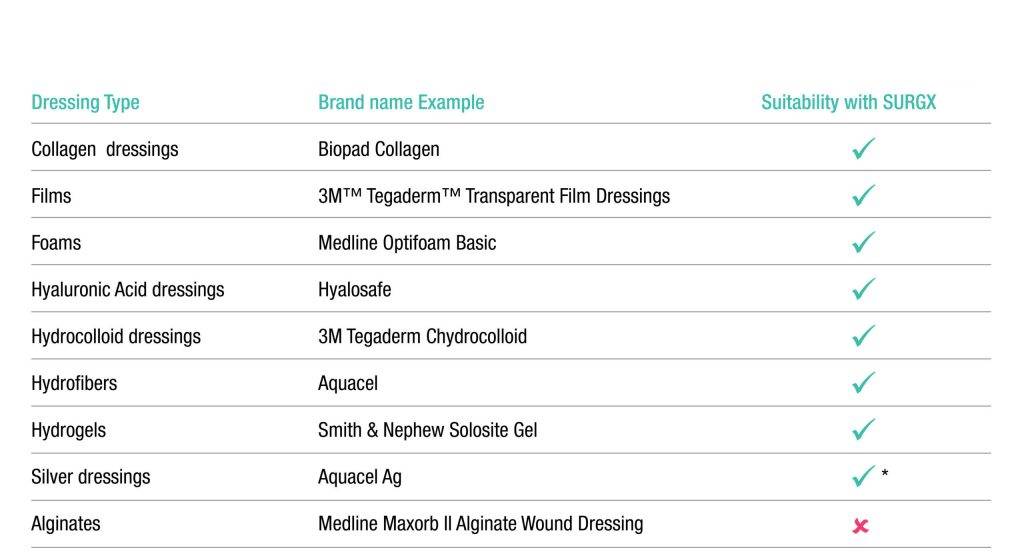
SURGX Sterile Antimicrobial Gel conforms to the postoperative wound. The gel provides a moist wound environment conducive to healing. While in place, the antimicrobial properties of the gel inhibit the growth of microorganisms in the product.
Interested in ordering?
Contact us at [email protected] or call (855) 968-6399