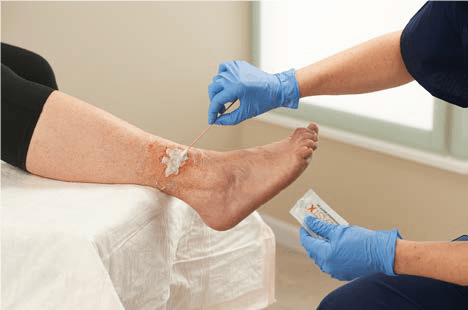
Start Your Healing Journey Day 1
Next Science’s BLASTX Antimicrobial Wound Gel provides wound management by maintaining a moist wound environment, which is conducive to wound healing. On day 1, the antimicrobial properties of the gel inhibit the growth of microorganisms in the product.